By Admin – Steroidal.com
Super DMZ Rx 2.0 – a legal steroid or designer steroid – can cause severe liver damage and jaundice, according to a study done in 2013 on an individual who ingested it.
Designer steroids, legal steroids and prohormones, often contain anabolic steroids, but their structural makeup has been altered slightly, so they don’t appear on the banned and prohibited dietary supplement items list the Drug Enforcement Agency (DEA) and Food and Drugs Administration (FDA) – overseas and enforces in the United States.
By reverse engineering illegal anabolic steroids and changing their composition, these drugs are permitted to be sold online and in retail stores as they’re not classified as illegal yet.
This loophole has been open for years, but that looks to change shortly as the Designer Anabolic Steroid Control Act (DASCA) has been passed by the House and is now edging closer and will become law in months to come. This new Bill will allow the DEA and FDA to add compounds and drugs to the banned list almost immediately, outlawing their sale, importation, manufacturer and possession in the USA.
 One popular designer steroid is – Super DMZ Rx 2.0 – manufactured by Blackstone Labs/Iron Mag Labs and sold online and in stores to the US market. This “dietary supplement” is advertised to “increase strength, power, lean, muscle gains and enhance fat loss” – but is actually a combination of two oral anabolic steroids, that can lead to side effects including liver damage.
One popular designer steroid is – Super DMZ Rx 2.0 – manufactured by Blackstone Labs/Iron Mag Labs and sold online and in stores to the US market. This “dietary supplement” is advertised to “increase strength, power, lean, muscle gains and enhance fat loss” – but is actually a combination of two oral anabolic steroids, that can lead to side effects including liver damage.
Super DMZ Rx 2.0 contains 10mg methylstenbolone and 10mg dymethazine, these are oral steroids, but not classified as illegal, meaning they can cause liver toxicity, kidney damage, adverse changes in cholesterol levels, acne, testosterone suppression, hair loss and virilisation changes in women.
Methylstenbolone is actually an effective oral anabolic steroid, as shown in numerous studies in the 1960’s on animals.
The second ingredient is dymethazine – which is actually a prohormone – converting to a more potent anabolic steroid after ingestion. After ingestion, dymethazine converts to two methasterone compounds. Methasterone is also an oral anabolic steroid but can cause liver stress when taken orally [1, 2, 3]. Dymethazine is closely linked to the #1 best selling prohormone – Superdrol.
According to a study done in 2013, by researchers at Ohio State University College of Medicine, a 26-year old man suffered severe liver damage after using Super DMZ Rx 2.0 [4].
The man took 2 caps of Super DMZ Rx 2.0 per day for a month, but noticed his urine had turned a darker colour during week 3. This is an indication of liver and/or kidney stress and often reported when users take high dosages of the injectable anabolic steroid – Trenbolone.
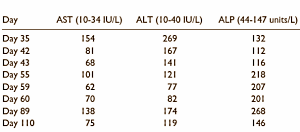
After becoming concerned, he stopped taking the designer steroid, but side effects didn’t subside. He then went to hospital, where doctors tested his liver enzymes – AST, ALT and ALP. The readings were high, but not as high as an alcoholic.
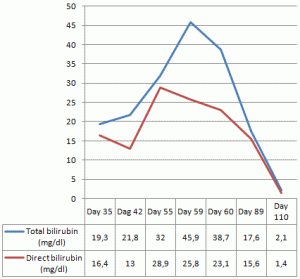
A liver biopsies was done, but again showed his readings weren’t alarmingly high, although they did find his direct bilirubin and total bilirubin were elevated.
Bilirubin is a waste product of haem, which is produced when the body is clearing up old red blood cells. This elevation shows liver damage from ingestion of the supplement.
The figure above shows that the man did recover after about 2 months of stopping Super DMZ Rx 2.0.
The scientists that covered this study express their concerns with designer steroid and prohormones sold labelled as “dietary supplements” and often users don’t comprehend the serious side effects that might experience from use.
“Although the manufacturer of Super DMZ Rx 2.0 did not conceal methylstenbolone and dymethazine as the major ingredients of the product, unregulated anabolic steroids use is unsafe. It is important for physicians to be aware of this while considering causes of jaundice since anabolic steroids induced liver injury can become fulminant,” the researchers concluded.
With the DASCA seeking to ban legal steroids like this, disguised as “dietary supplements” seeing reports like this should become rare.
References:
[1]. Clin Gastroenterol Hepatol. 2008 Feb;6(2):255-8. doi: 10.1016/j.cgh.2007.11.010. Epub 2008 Jan 9.
[2]. Clin Gastroenterol Hepatol. 2007 Jul;5(7):809-12. Epub 2007 May 16.
[3]. Am J Gastroenterol. 2006 Nov;101(11):2659-62. Epub 2006 Sep 4.
[4]. Journal of Investigative Medicine High Impact Case Reports April-June 2014vol. 2 no. 2 2324709614532800